Pivotal Trial Demonstrated Superior Acute and Long-Term Patency Over Provisional Stenting
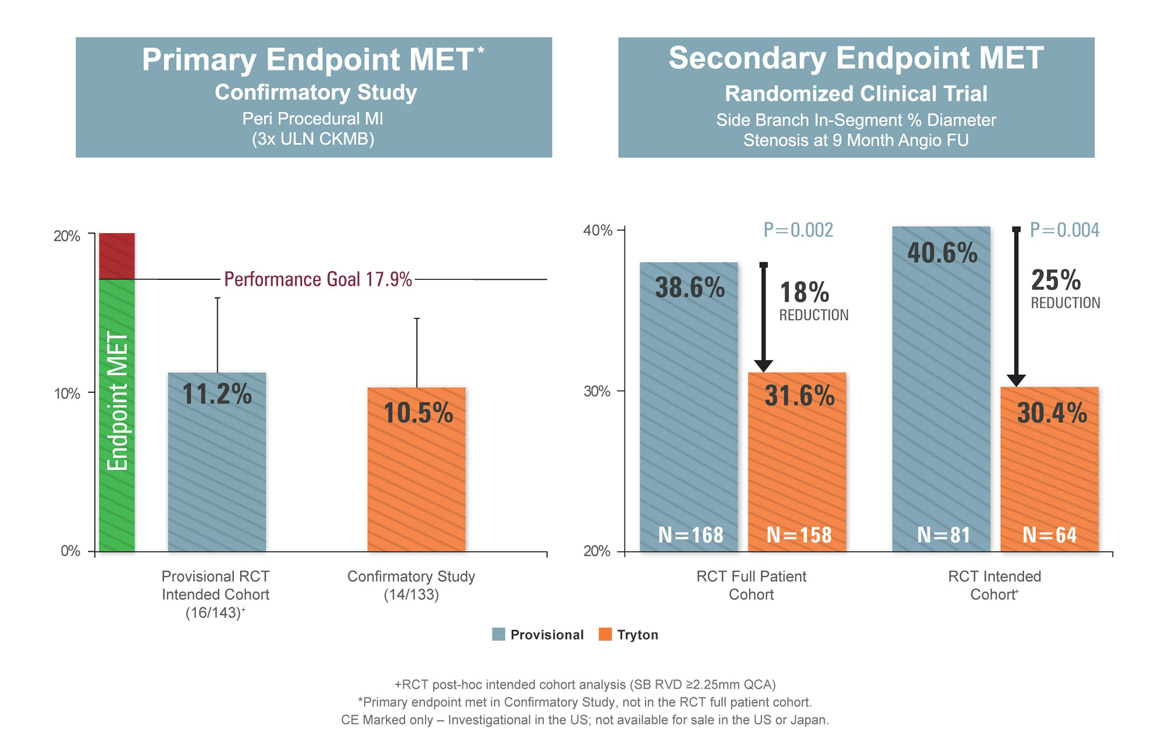
The landmark Tryton IDE study was a multi-national randomized trial that compared a Tryton stent in the side branch vs. the use of balloon angioplasty in the side branch, with both arms of the trial utilizing a standard drug eluting stent in the main vessel. The study, which was the first and only randomized IDE clinical trial of a bifurcation stent, enrolled over 700 patients from up to 75 centers in North America, Europe and Israel. Martin Leon, M.D. (Columbia University, New York) served as principal investigator for the study and Patrick Serruys, M.D., PhD. (Thoraxcenter, Rotterdam) led the IVUS.
Publications
Dedicated Bifurcation Stent for the Treatment of Bifurcation Lesions Involving Large Side Branches
Outcomes of Tryton in Bifurcations with Large Side Branches – RCT Post Hoc Analysis
Tryton Randomized Trial
6M And 1Y Clinical Outcomes: A Patient-Level Pooled Analysis Of 8 Registry Studies
Poster Abstracts
2-Year Clinical Follow-up of the Tryton IDE Randomized Trial
Stenting of Coronary Bifurcation Lesions with Bioresorbable Everoliumus- Eluting Scaffolds
Incidence of Peri-Procedural MI: Insight from the Tryton IDE Randomized Trial
TCT 2016 Symposium Presentations
The following presentations represent external content and are being provided as a convenience and for informational purposes only; they do not constitute an endorsement or an approval by Tryton Medical for any of the statements or opinions of the individual or organization.
TCT 2015 Symposium Presentations
Stent Expansion and Lesion Coverage Insights From Intravascular Imaging | Antonio L. Bartorelli, M.D.
Lesion Coverage: Insights from the Bench Top | Maciej Lesiak, M.D.
Tryton – Addressing the Challenge of Bifurcation Stenting | Dean J. Kereiakes, M.D.
Dedicated Bifurcation Stent Technology: Implications for Everyday Practice | Jens Flensted Lassen, M.D.
Bifurcation Lesion Treatment Option: Insights From Clinical Literature | James Hermiller, M.D.
Tryton Pivotal: Randomized Trial and Confirmatory Study - Key Messages | Martin B. Leon, M.D.
The Tryton Pivotal: Randomized Trial & Confirmatory Study | Philippe Généreux, M.D.






